End-to-End Solution for Magnetization Automation
Automated Magnetic Charging Integrated Solution
Formaldehyde (HCHO) is a widespread water-soluble toxic pollutant, which mainly originates from industrial production emissions, volatilization of building and decoration materials, and the release of daily necessities. This compound is strongly carcinogenic and biotoxic, which will not only cause serious damage to the human respiratory system and immune system, but also destroy the ecological balance of the water body. Therefore, the development of efficient formaldehyde removal technology is of great significance for the protection of human health and the maintenance of ecosystem stability. The existing formaldehyde degradation methods mainly include adsorption technology, biodegradation method and catalytic oxidation method, among which catalytic oxidation method has become the key direction of the current research due to its advantages of complete degradation and no secondary pollution.
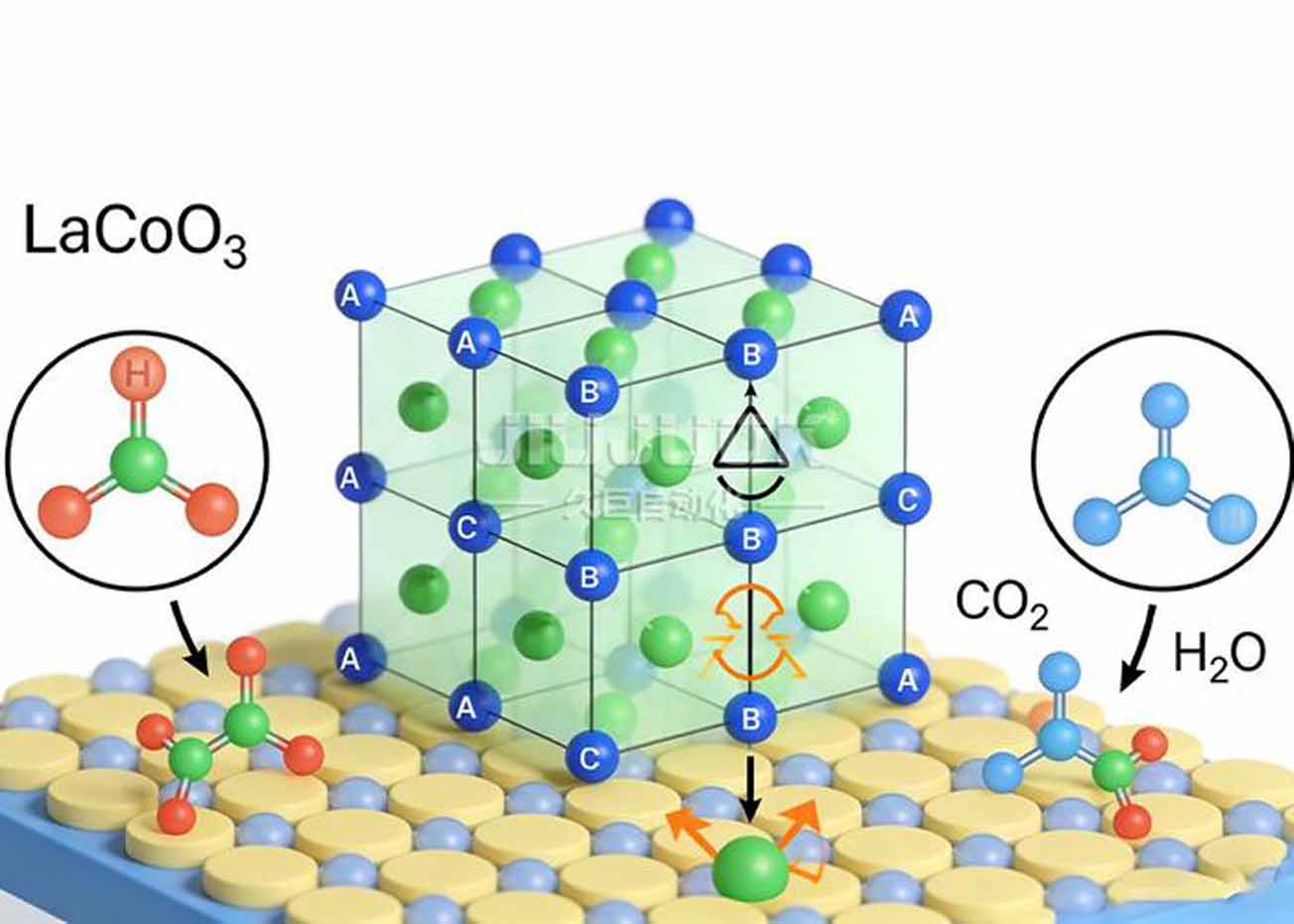
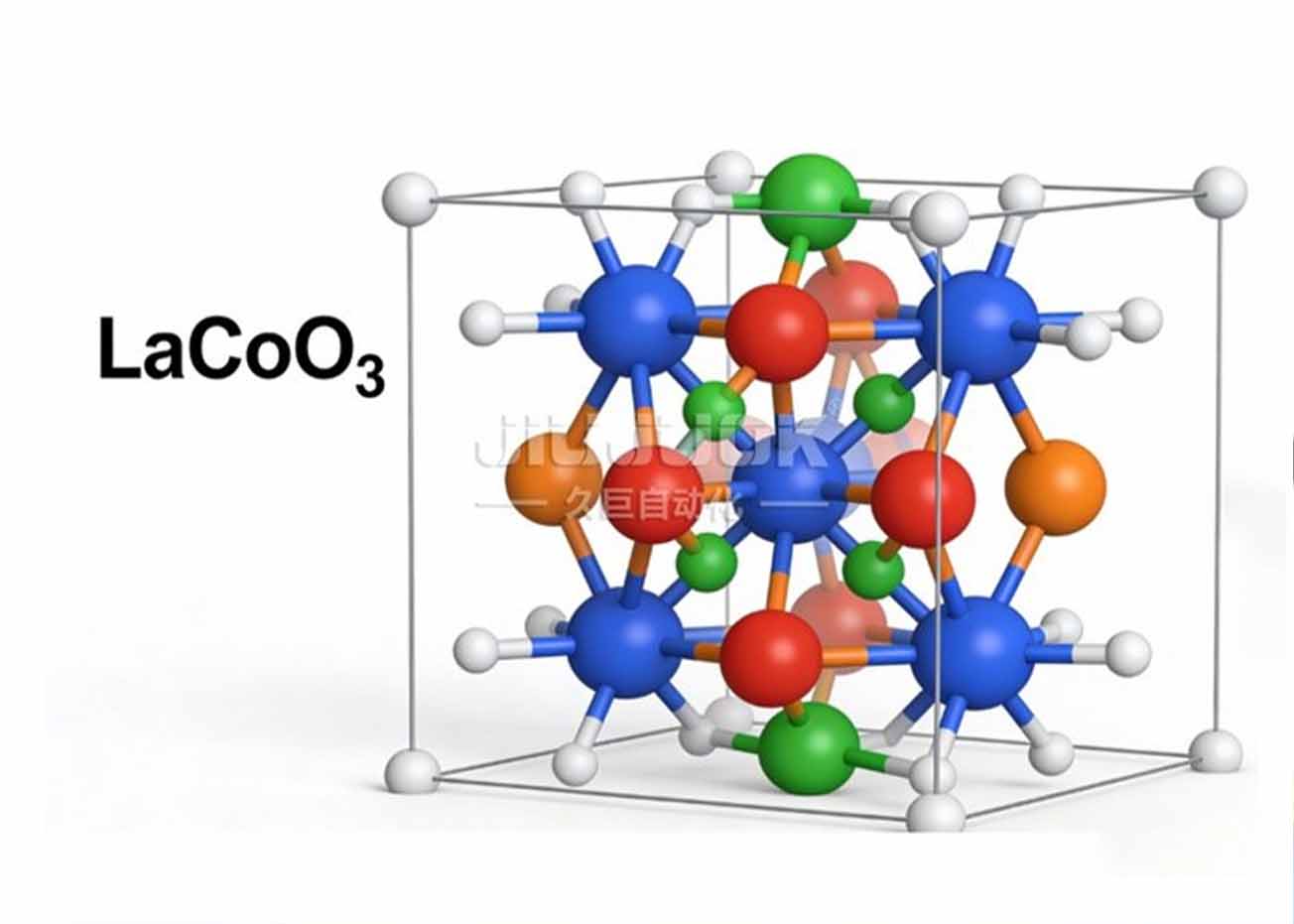
Calcite-type oxides (ABO₃), with unique crystal structures, excellent electronic conductivity and stable catalytic activity, have made remarkable progress in multiphase catalysis such as CO oxidation and volatile organic compound degradation. As a typical chalcogenide material, lanthanum cobalt oxide (LaCoO₃) has a potential oxidation catalytic ability due to the valence tunability of its active component cobalt (Co). However, there are relatively few studies on the degradation of formaldehyde in water by LaCoO₃, and the mechanism of its catalytic activity and reaction pathway are not yet fully understood.
Based on the above background, in this study, chalcogenide LaCoO₃ catalysts with different lanthanum to cobalt molar ratios were prepared by the sol-gel method, and the catalytic degradation of formaldehyde in water was systematically investigated at room temperature, and the catalytic mechanism was also revealed by the combination of the material characterization technique and the free radical burst experiments. The results of catalytic performance tests showed that the prepared LaCoO₃ catalysts exhibited excellent formaldehyde degradation activity. Compared with the traditional non-homogeneous catalysts (e.g., transition metal oxides, noble metal-loaded catalysts, etc.), the formaldehyde degradation time was shortened from 119 min to 10 min, and the degradation efficiency was improved by 12 times. The best degradation activity was achieved when the lanthanum to cobalt molar ratio was 1:1, and the complete removal of formaldehyde from water could be realized in 10 min at room temperature, and the degradation rate remained above 95% after 3 times of reuse, which showed good stability.
In order to elucidate the intrinsic reasons for the differences in catalytic activity, the surface electronic states of the catalysts with different lanthanum-cobalt molar ratios were analyzed in this study using X-ray photoelectron spectroscopy (XPS). The results showed that the relative content of Co²⁺ on the catalyst surface gradually decreased with the increase of lanthanum-cobalt molar ratio, while the content of Co³⁺ increased accordingly. Combined with the catalytic performance data, it can be found that the formaldehyde degradation activity of the catalyst is positively correlated with the Co²⁺ content, indicating that Co²⁺ is the key active site to promote the oxidative degradation of formaldehyde, and participates in the redox reaction through the Co²⁺/Co³⁺ valence cycle. reaction, accelerating the generation of active species.
In order to identify the main active species during the reaction process, this study further conducted free radical bursting experiments by adding sulfate radical (SO₄・-) bursting agent (anhydrous ethanol) and hydroxyl radical (・OH) bursting agent (tert-butanol) to the reaction system, respectively. The experimental results showed that the addition of both bursting agents significantly inhibited the degradation efficiency of formaldehyde, in which the degradation rate decreased by 681 TP3T with the addition of anhydrous ethanol and 521 TP3T with the addition of tert-butanol, which confirms that SO₄・- and ・OH are the main active species in the degradation of formaldehyde. Based on the above results, the present study proposed that the reaction mechanism of LaCoO₃-catalyzed formaldehyde degradation is a non-homogeneous Fenton-like oxidation reaction: Co²⁺ on the catalyst surface reacts with oxidants in the system (e.g., dissolved oxygen, hydrogen peroxide, etc.) to form SO₄・- and・OH, which then oxidizes formaldehyde into CO₄ and・OH through strong oxidative action. Formaldehyde is oxidized and decomposed into CO₂ and H₂O.
In this study, the potential application of chalcogenide LaCoO₃ in formaldehyde degradation in water was confirmed for the first time, the mechanism of lanthanide-cobalt molar ratio on the catalytic activity was clarified, and the reaction pathway of non-homogeneous Fenton-like oxidation was revealed, which provided the theoretical basis and technical reference for the development of highly efficient and stable catalysts for formaldehyde degradation at room temperature.
Magnetization of Magnetic Materials
3C Electronic Magnetization
Motor Rotor Magnetization
Car Speaker Magnetization
New Energy Vehicle Industry
Medical Equipment Industry

February 25, 2026
Automated Magnetic Charging Integrated Solution Magnetic Charging Equipment Magnetic Charging Automation Magnetic Measuring Instruments Magnetic Charging Service In the industrial manufacturing field, silicone magnetic products are widely used in smart wear and sealing components because of their flexibility and magnetic properties. The core of their magnetic properties depends on professional magnetization process. This time, JiuJu magnetization undertakes the project of magnetizing silicone products, and successfully realizes the product's magnetic force from 0.01 gauss to 15.5 gauss by leaps and bounds, the following is the whole process detail...

January 7, 2026
Automated Magnetic Charging Integrated Solution Magnetic Charging Equipment Magnetic Charging Automation Magnetic Measuring Instruments Magnetic Charging Services Magnetic stripes are widely used in medical cards, medical insurance cards and other medical credentials to store key data such as basic patient information and medical insurance participation information. Due to the frequency of use and the external environment, the magnetic stripe may become magnetically weakened or demagnetized, resulting in the inability of the equipment to read the information, and then need to be magnetized. The core principle of magnetization is to make the magnetic strip inside the...

January 6, 2026
Automated Magnetic Charging Integrated Solution Magnetic Charging Equipment Magnetic Charging Automation Magnetic Measuring Instruments Magnetic Charging Service This paper focuses on the core process of magnetization of Rubber Silicone and NdFeB, the initial state of non-magnetic before magnetization, and the magnetic performance after magnetization. Through the gaussmeter measurement verification, it is clear that the highest magnetic unit after magnetization is up to 45.9 units and the highest magnetic unit after magnetization is up to 45.9 units (measured by gaussmeter). The magnetization process is the core link to realize the magnetic transformation of the two...

November 13, 2025
Specializing in ferrite cube micro-magnetization, we deliver customized, precise solutions tailored to ferrite cube material properties. By fine-tuning parameters, we ensure the magnetization meets exact standards, fulfilling high-precision magnetic requirements and adapting to applications with strict demands for magnetization accuracy.

2026年4月11日
Automated Magnetic Charging Integrated Solution 充磁设备 充磁自动化 磁测量仪器 充磁服务 选购充磁机切勿只以价格为导向,核心应围绕设备关键参数与实际生产场景综合考量。重点关注充磁电压、输出功率、电容容量等核心配置,同时结合所充磁材类型、尺寸规格及生产节拍进行匹配选型。若参数与需求不匹配,极易出现充磁饱和度不足、磁场分…

2026年4月10日
Automated Magnetic Charging Integrated Solution 充磁设备 充磁自动化 磁测量仪器 充磁服务 面对多型号电机充磁选型难题,久巨电机充磁设备以高兼容性核心设计,精准破解生产痛点。设备彻底打破传统 “一机一用” 模式,采用 “更换治具 + 调试程序” 的灵活切换方案,无需整机改造,即可快速适配不同尺寸、极数及充磁方向的电机产品。操作简…

February 27, 2026
JiuJu Magnetization Case - Antique Audio Speaker Magnetization Restoration, Technology and Example Introduction

February 26, 2026
Automated Magnetic Charging Integrated Solution Magnetizing Equipment Magnetizing Automation Magnetic Measuring Instruments Magnetizing Service Magnetizing is the core step in the manufacture and maintenance of permanent magnet motors such as permanent magnet synchronous motors (PMS) and brushless DC motors (BDC). It relies on the principle of electromagnetic induction and magnetic field energy conversion, with the help of external strong magnetic field to make the internal magnetic domains of permanent magnets arranged in a directional manner, so as to form stable and long-lasting magnetism, which is the basic link to ensure the normal operation of the motor. First, the essence of magnetization: to give the motor...

2026年4月11日
Automated Magnetic Charging Integrated Solution 充磁设备 充磁自动化 磁测量仪器 充磁服务 选购充磁机切勿只以价格为导向,核心应围绕设备关键参数与实际生产场景综合考量。重点关注充磁电压、输出功率、电容容量等核心配置,同时结合所充磁材类型、尺寸规格及生产节拍进行匹配选型。若参数与需求不匹配,极易出现充磁饱和度不足、磁场分…

2026年4月10日
Automated Magnetic Charging Integrated Solution 充磁设备 充磁自动化 磁测量仪器 充磁服务 面对多型号电机充磁选型难题,久巨电机充磁设备以高兼容性核心设计,精准破解生产痛点。设备彻底打破传统 “一机一用” 模式,采用 “更换治具 + 调试程序” 的灵活切换方案,无需整机改造,即可快速适配不同尺寸、极数及充磁方向的电机产品。操作简…

February 27, 2026
JiuJu Magnetization Case - Antique Audio Speaker Magnetization Restoration, Technology and Example Introduction

February 26, 2026
Automated Magnetic Charging Integrated Solution Magnetizing Equipment Magnetizing Automation Magnetic Measuring Instruments Magnetizing Service Magnetizing is the core step in the manufacture and maintenance of permanent magnet motors such as permanent magnet synchronous motors (PMS) and brushless DC motors (BDC). It relies on the principle of electromagnetic induction and magnetic field energy conversion, with the help of external strong magnetic field to make the internal magnetic domains of permanent magnets arranged in a directional manner, so as to form stable and long-lasting magnetism, which is the basic link to ensure the normal operation of the motor. First, the essence of magnetization: to give the motor...

2026年4月11日
Automated Magnetic Charging Integrated Solution 充磁设备 充磁自动化 磁测量仪器 充磁服务 选购充磁机切勿只以价格为导向,核心应围绕设备关键参数与实际生产场景综合考量。重点关注充磁电压、输出功率、电容容量等核心配置,同时结合所充磁材类型、尺寸规格及生产节拍进行匹配选型。若参数与需求不匹配,极易出现充磁饱和度不足、磁场分…

2026年4月10日
Automated Magnetic Charging Integrated Solution 充磁设备 充磁自动化 磁测量仪器 充磁服务 面对多型号电机充磁选型难题,久巨电机充磁设备以高兼容性核心设计,精准破解生产痛点。设备彻底打破传统 “一机一用” 模式,采用 “更换治具 + 调试程序” 的灵活切换方案,无需整机改造,即可快速适配不同尺寸、极数及充磁方向的电机产品。操作简…

February 27, 2026
JiuJu Magnetization Case - Antique Audio Speaker Magnetization Restoration, Technology and Example Introduction

February 26, 2026
Automated Magnetic Charging Integrated Solution Magnetizing Equipment Magnetizing Automation Magnetic Measuring Instruments Magnetizing Service Magnetizing is the core step in the manufacture and maintenance of permanent magnet motors such as permanent magnet synchronous motors (PMS) and brushless DC motors (BDC). It relies on the principle of electromagnetic induction and magnetic field energy conversion, with the help of external strong magnetic field to make the internal magnetic domains of permanent magnets arranged in a directional manner, so as to form stable and long-lasting magnetism, which is the basic link to ensure the normal operation of the motor. First, the essence of magnetization: to give the motor...

2026年4月11日
Automated Magnetic Charging Integrated Solution 充磁设备 充磁自动化 磁测量仪器 充磁服务 选购充磁机切勿只以价格为导向,核心应围绕设备关键参数与实际生产场景综合考量。重点关注充磁电压、输出功率、电容容量等核心配置,同时结合所充磁材类型、尺寸规格及生产节拍进行匹配选型。若参数与需求不匹配,极易出现充磁饱和度不足、磁场分…

2026年4月10日
Automated Magnetic Charging Integrated Solution 充磁设备 充磁自动化 磁测量仪器 充磁服务 面对多型号电机充磁选型难题,久巨电机充磁设备以高兼容性核心设计,精准破解生产痛点。设备彻底打破传统 “一机一用” 模式,采用 “更换治具 + 调试程序” 的灵活切换方案,无需整机改造,即可快速适配不同尺寸、极数及充磁方向的电机产品。操作简…

February 27, 2026
JiuJu Magnetization Case - Antique Audio Speaker Magnetization Restoration, Technology and Example Introduction

February 26, 2026
Automated Magnetic Charging Integrated Solution Magnetizing Equipment Magnetizing Automation Magnetic Measuring Instruments Magnetizing Service Magnetizing is the core step in the manufacture and maintenance of permanent magnet motors such as permanent magnet synchronous motors (PMS) and brushless DC motors (BDC). It relies on the principle of electromagnetic induction and magnetic field energy conversion, with the help of external strong magnetic field to make the internal magnetic domains of permanent magnets arranged in a directional manner, so as to form stable and long-lasting magnetism, which is the basic link to ensure the normal operation of the motor. First, the essence of magnetization: to give the motor...

2026年4月11日
Automated Magnetic Charging Integrated Solution 充磁设备 充磁自动化 磁测量仪器 充磁服务 选购充磁机切勿只以价格为导向,核心应围绕设备关键参数与实际生产场景综合考量。重点关注充磁电压、输出功率、电容容量等核心配置,同时结合所充磁材类型、尺寸规格及生产节拍进行匹配选型。若参数与需求不匹配,极易出现充磁饱和度不足、磁场分…

2026年4月10日
Automated Magnetic Charging Integrated Solution 充磁设备 充磁自动化 磁测量仪器 充磁服务 面对多型号电机充磁选型难题,久巨电机充磁设备以高兼容性核心设计,精准破解生产痛点。设备彻底打破传统 “一机一用” 模式,采用 “更换治具 + 调试程序” 的灵活切换方案,无需整机改造,即可快速适配不同尺寸、极数及充磁方向的电机产品。操作简…

February 27, 2026
JiuJu Magnetization Case - Antique Audio Speaker Magnetization Restoration, Technology and Example Introduction

February 26, 2026
Automated Magnetic Charging Integrated Solution Magnetizing Equipment Magnetizing Automation Magnetic Measuring Instruments Magnetizing Service Magnetizing is the core step in the manufacture and maintenance of permanent magnet motors such as permanent magnet synchronous motors (PMS) and brushless DC motors (BDC). It relies on the principle of electromagnetic induction and magnetic field energy conversion, with the help of external strong magnetic field to make the internal magnetic domains of permanent magnets arranged in a directional manner, so as to form stable and long-lasting magnetism, which is the basic link to ensure the normal operation of the motor. First, the essence of magnetization: to give the motor...
 Jiu Ju Magnetizer
Jiu Ju Magnetizer